:max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
The details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements in the Periodic Table. Neon has the Atomic Mass weight of 20.18. The number of protons in atom of neon is therefore 10. Example 3 - mass number of Neon: The element Neon (Symbol Ne) has the Atomic Number of 10.The periodic table is arguably the best tool in all of science no other branch of science can. The periodic table lists all the known chemical elements, arranged by atomic number (that is, the number of protons in the nucleus). In this chapter, we present some data on the chemical elements. The mass number of silver is therefore 108. Appendix: Periodic Table of the Elements. Silver has the Atomic Mass weight of 107.87. The number of protons in atom of silver is therefore 47. Example 2 - mass number of Silver: The element Silver (Symbol Ag) has the Atomic Number of 47.The mass number of gold is therefore 197. Gold has the Atomic Mass weight of 196.97. The number of protons in atom of gold is therefore 79. Example 1 - mass number of Gold: The element Gold (Symbol Au) has the Atomic Number of 79.The following examples provide details of how to calculate the mass number. The unique chart below has been created by and details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements. According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. So, if we know the number of protons and neutrons in an atom we can determine the mass number. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Mass Numbers - The Mass Numbers of all of the elements 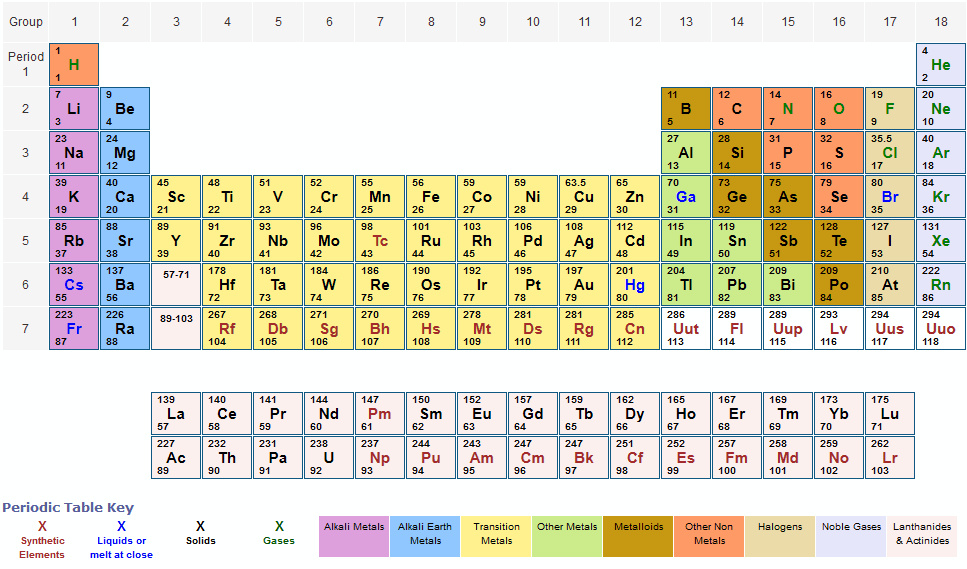
Number of Neutrons = Mass Number - Atomic Number Mass Numbers Mass Numbers = Atomic Weight of Element, rounded to nearest whole number Subtracting the Atomic number from the Mass Number equals the number of neutrons in the nucleus. Mass numbers equal the total number of heavy, or massive, particles in the nucleus. The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom. The chemical properties of an element are determined by its Atomic Number not its Mass Number which is why atomic numbers are shown on the Periodic table whilst Mass Numbers are not. The Periodic Table with Atomic Mass will give you the atomic weight, or atomic mass, of the elements. The accompanying table lists some useful hydrates.The mass number is established by rounding the atomic weight to the nearest whole number. Aluminum chloride hexahydrate is an active ingredient in antiperspirants. Epsom salt (MgSO 4♷H 2O) is used as a bathing salt and a laxative. ½H 2O), known as plaster of Paris, is used to make casts for broken bones. Periodic Table Of Elements Rounded Atomic Mass The Regular Table is a crucial part of the research into scientific research, also it can be useful in discovering a substance’s qualities.Hydrates have various uses in the health industry. The name of this compound is copper(II) sulfate pentahydrate, with the penta- prefix indicating the presence of five water units per formula unit of copper(II) sulfate. The hydrate of copper(II) sulfate has five water units associated with each formula unit, so it is written as CuSO 4♵H 2O. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The two chemical formulas are separated by a vertically centered dot. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. To write the chemical formula of a hydrate, write the number of water units per formula unit of compound after its chemical formula. Hydrates are solids, not liquids or solutions, despite the water they contain. These compounds, called hydrates, have a characteristic number of water units associated with each formula unit of the compound. Some ionic compounds have water (H 2O) incorporated within their formula unit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
